The retina is an important part of the eye, as it converts light into electrical signals that are later processed in the brain. It acts as a biological photodetector. Imaging the structure and function of the living retina is crucial for effectively diagnosing and treating eye diseases and drug development. Structural information about the retina can be obtained through OCT studies, among others. However, functional changes are the first signs of early pathological processes and often precede structural changes; obtaining this information is currently very difficult. ICTER researchers are working on a new method for functional imaging of the fundus based on two-photon excited fluorescence.
The retina has a layered structure that is filled with various fluorophores. For example, the retinal pigment epithelium (RPE) contains lipofuscin, a byproduct of the visual cycle. Lipofuscin accumulates with age but also as the disease progresses. Other examples include retinol and retinyl esters (vitamin A derivatives active in the visual cycle), melanin, FAD, NADH, collagen, and elastin. These substances can provide valuable information about the retina’s health and can be a valuable tool for detecting functional changes in age-related macular degeneration, diabetic retinopathy, or glaucoma.
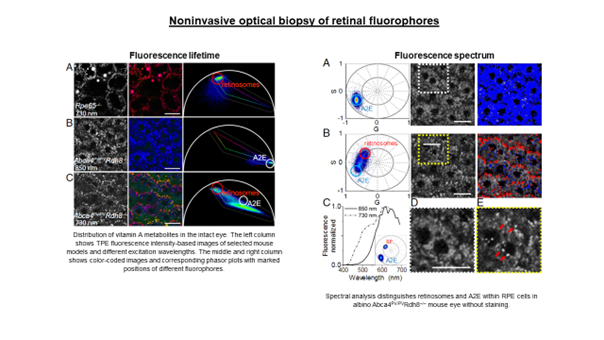
Standard ocular autofluorescence imaging visualizes the distribution of retinal fluorophores, but only intensity information is available. As a result, signals from different fluorophores cannot be distinguished. Lipofuscin is the dominant fluorophore, and its strong signal is mixed with others, usually from much weaker sources. Different fluorophores differ in their fluorescence properties, i.e., fluorescence lifetime and fluorescence spectrum. This provides an additional discriminating parameter to distinguish them from each other.
The eye is a window to the world but has a certain transmission range. Consequently, many fluorophores with excitation spectra in the UV/blue range (<420 nm) cannot be excited, and the information they contain is unavailable. Our solution to this problem is to use two-photon excitation. This scheme uses short (femtosecond) pulses in the near-infrared (twice the wavelength), which bypasses the limitations due to the transmission of the eye. For example, the use of femtosecond pulses at 730 nm is equivalent to single-photon excitation at 365 nm, which would not be possible in the living eye. Additional advantages of this method include better resolution, less phototoxicity, and less scattering.
In our research, we aim to visualize eye tissue structure and molecular composition using spectral and temporal discrimination. For example, the image below shows the fluorescence lifetime distribution (FLIM) of retinal pigment epithelial cells of Abca4PV/PV mice (a model of Stargardt disease in humans). The image reflects differences in the pigment epithelium’s subcellular distribution of endogenous fluorophores. Shorter lifetimes (blue-green color) are associated with A2E, a component of lipofuscin. Red granules (longer lifetimes) may be associated with retinyl esters.
Author: Jakub Bogusławski, PhD jboguslawski@ichf.edu.pl
Team: Grażyna Palczewska, Jakub Bogusławski, Łukasz Kornaszewski, Maciej Wojtkowski
Publication:
Grazyna Palczewska, Jakub Boguslawski, Patrycjusz Stremplewski, Lukasz Kornaszewski, Jianye Zhang, Zhiqian Dong, Xiao-Xuan Liang, Enrico Gratton, Alfred Vogel, Maciej Wojtkowski, Krzysztof Palczewski, “Noninvasive two-photon optical biopsy of retinal fluorophores,” Proceedings of the National Academy of Sciences 117(36), 22532-22543 (2020).

